What GMP Certification Means for Supplement Quality
GMP certification demonstrates that a third-party auditor has verified a manufacturer's quality management systems meet defined standards at the time of audit. This certification confirms documented processes for ingredient testing, production controls, deviation investigation, and record maintenance—all critical elements for consistent manufacturing. The distinction between mandatory cGMP compliance (required by FDA under 21 CFR Part 111) and voluntary third-party GMP certification is crucial: all registered U.S. manufacturers must comply with cGMP regulations, but certification from organizations like NSF, USP, or NPA provides independent verification of that compliance.
Testing Requirements Under 21 CFR Part 111
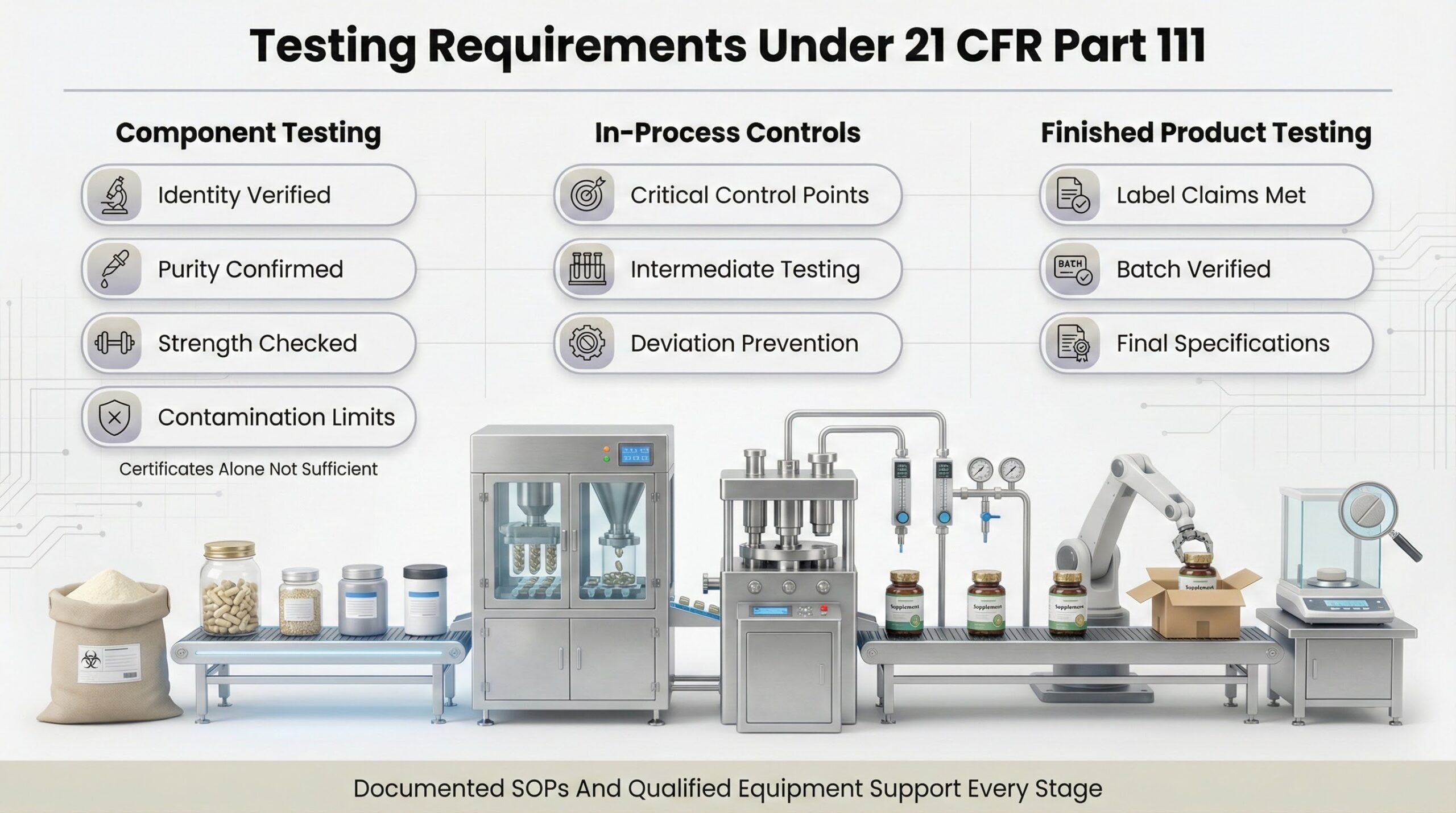
The cGMP framework requires manufacturers to establish specifications and conduct testing at multiple stages. Component specifications must verify identity, purity, strength, composition, and contamination limits for incoming dietary ingredients—certificates of analysis alone are insufficient. In-process testing at critical control points ensures intermediate materials meet standards before they compromise finished products. Finished product testing verifies each batch meets label claims and specifications for identity, strength, purity, and composition. Standard operating procedures must be documented for all production steps, and manufacturing equipment must be qualified and maintained to prevent contamination.

Why Certification Doesn't Guarantee Safety
Despite GMP standards, significant quality problems persist in the supplement industry. A 2011 study found that 73% of supplement manufacturers inspected by the FDA violated at least one GMP regulation. Contamination remains a persistent concern: a 2010 Government Accountability Office analysis found that 93% of supplements tested contained trace amounts of lead, arsenic, mercury, cadmium, or pesticides. Research published in JAMA found that 20.7% of Ayurvedic medicines purchased online contained detectable lead, mercury, or arsenic, with 75% of metal-containing products claiming Good Manufacturing Practices. All metal-containing products in that study exceeded at least one regulatory standard for acceptable daily metal intake.

The Efficacy Gap
GMP certification addresses manufacturing consistency but says nothing about whether a supplement actually works. NSF certification explicitly does not test for efficacy—it verifies that products contain what the label claims and are produced safely, but makes no assessment of biological effectiveness. This represents a fundamental limitation: a product can be manufactured perfectly to specification while delivering no meaningful health benefit. Stability testing evaluates how environmental factors affect quality over time and helps determine shelf life, but bioavailability testing—which would assess how well active ingredients are absorbed and utilized—is not mandated under 21 CFR Part 111.

Manufacturing Method Matters for Consistency
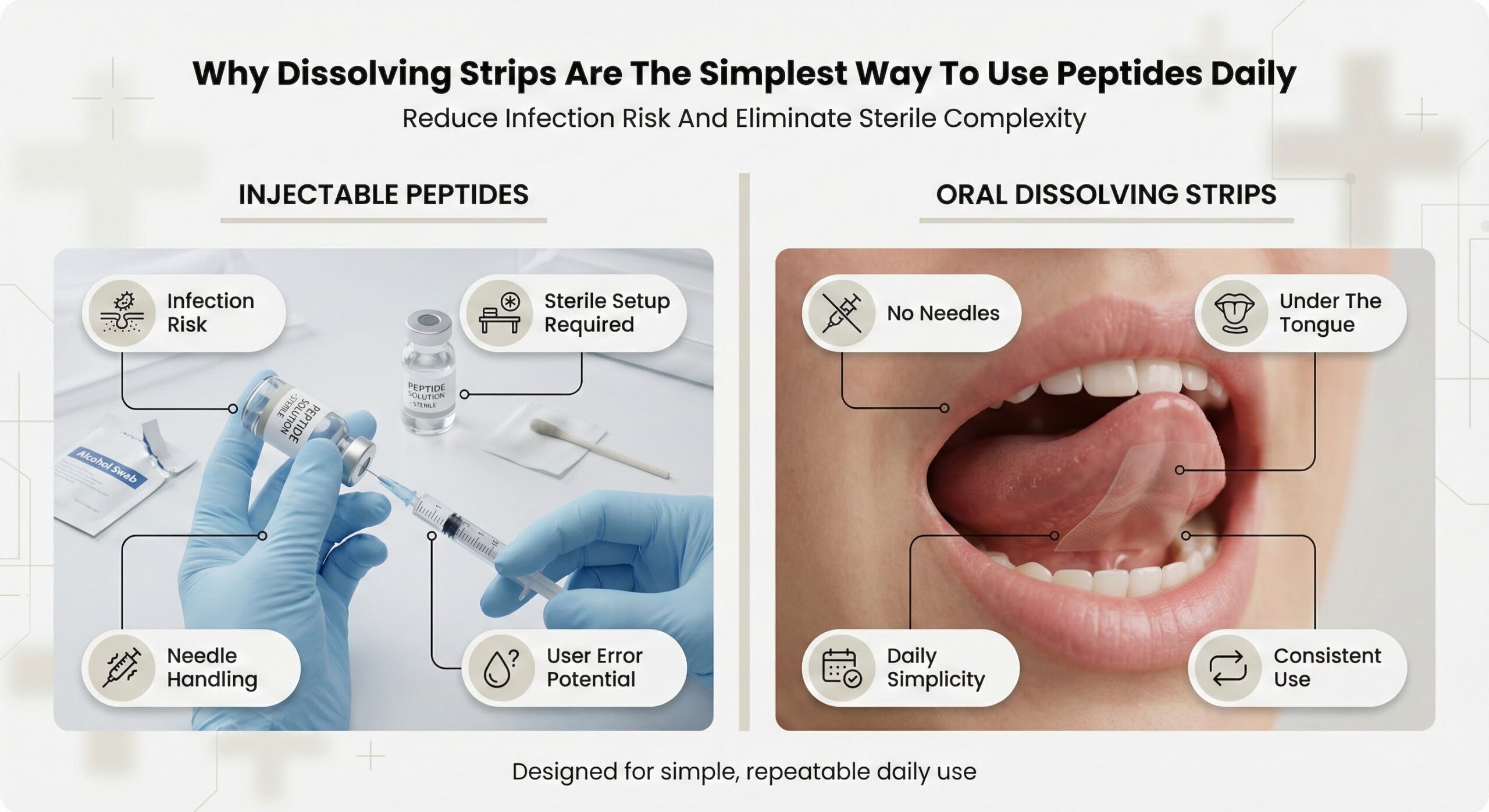
The reliability of supplement benefits depends not just on ingredient quality but on consistent, controlled production that minimizes user error and variability. GMP-certified facilities must document master manufacturing records, maintain batch-to-batch consistency, and implement process controls that ensure every product meets pre-set specifications. For consumers seeking peptide benefits, delivery systems that reduce preparation complexity and measurement error increase the likelihood of achieving consistent outcomes over time. Oral dissolving strips manufactured under GMP certification eliminate reconstitution steps, injection-site variables, and dosing measurement that introduce variability in traditional delivery methods.[1]

What Certification Actually Proves
GMP certification verifies that quality systems exist and function at the moment of audit—it demonstrates repeatable processes, documented controls, supplier qualification, complaint handling, and recall procedures. However, certification does not constitute FDA approval, does not guarantee future compliance without ongoing surveillance, and does not eliminate contamination risk, particularly for botanical ingredients prone to environmental contamination. For meaningful quality assurance, GMP certification should be combined with third-party product testing that independently verifies finished products for label accuracy, purity, and contaminant limits.
References
- Bala R et al. "Orally dissolving strips: A new approach to oral drug delivery system." Int J Pharm Investig. 2013. [View Study]
- Starr RR "Too little, too late: ineffective regulation of dietary supplements in the United States." Am J Public Health. 2015. [View Study]
- [View Study]
Read More
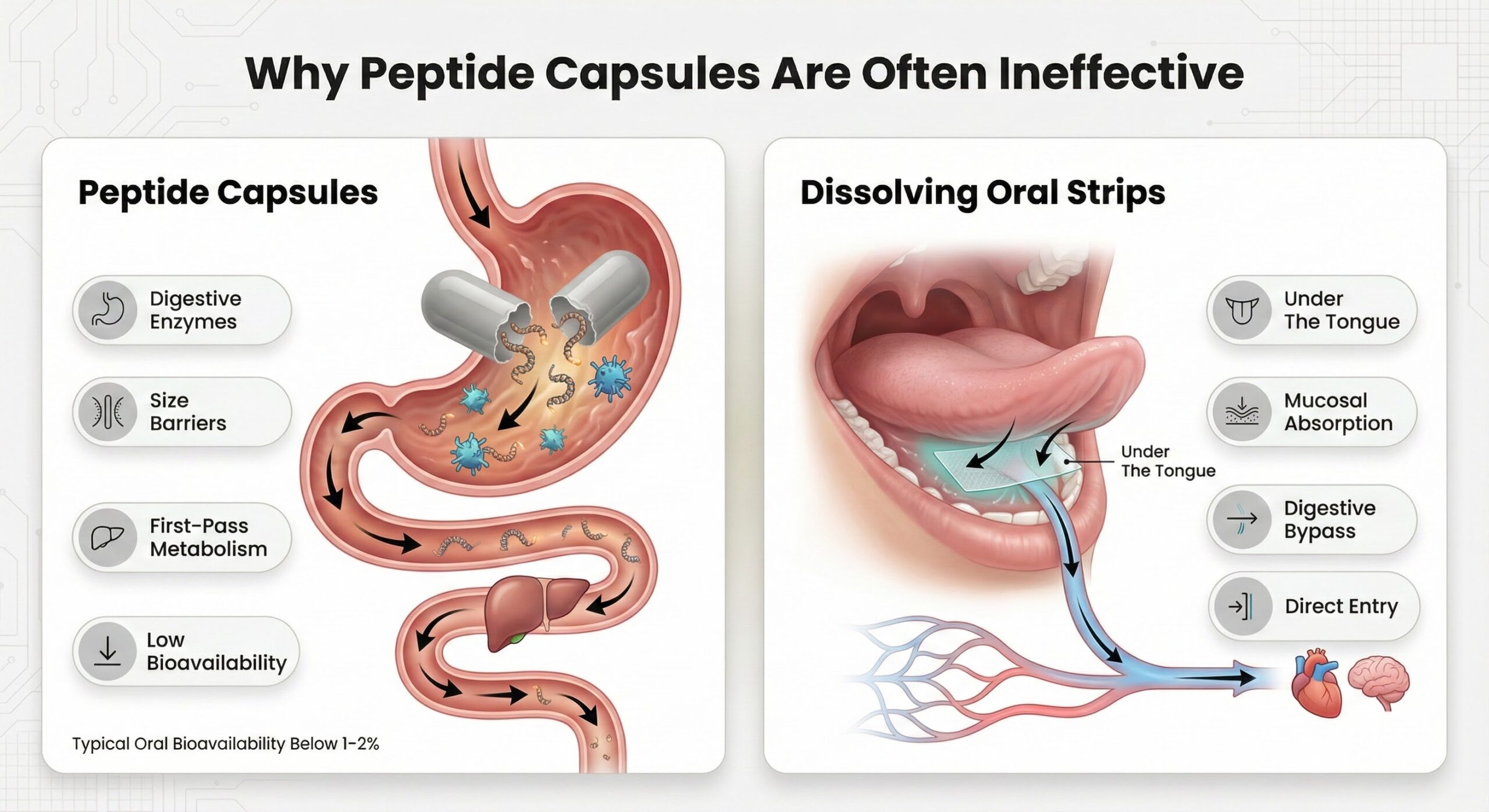
Why Peptide Capsules are Often Ineffective
March 10, 2026
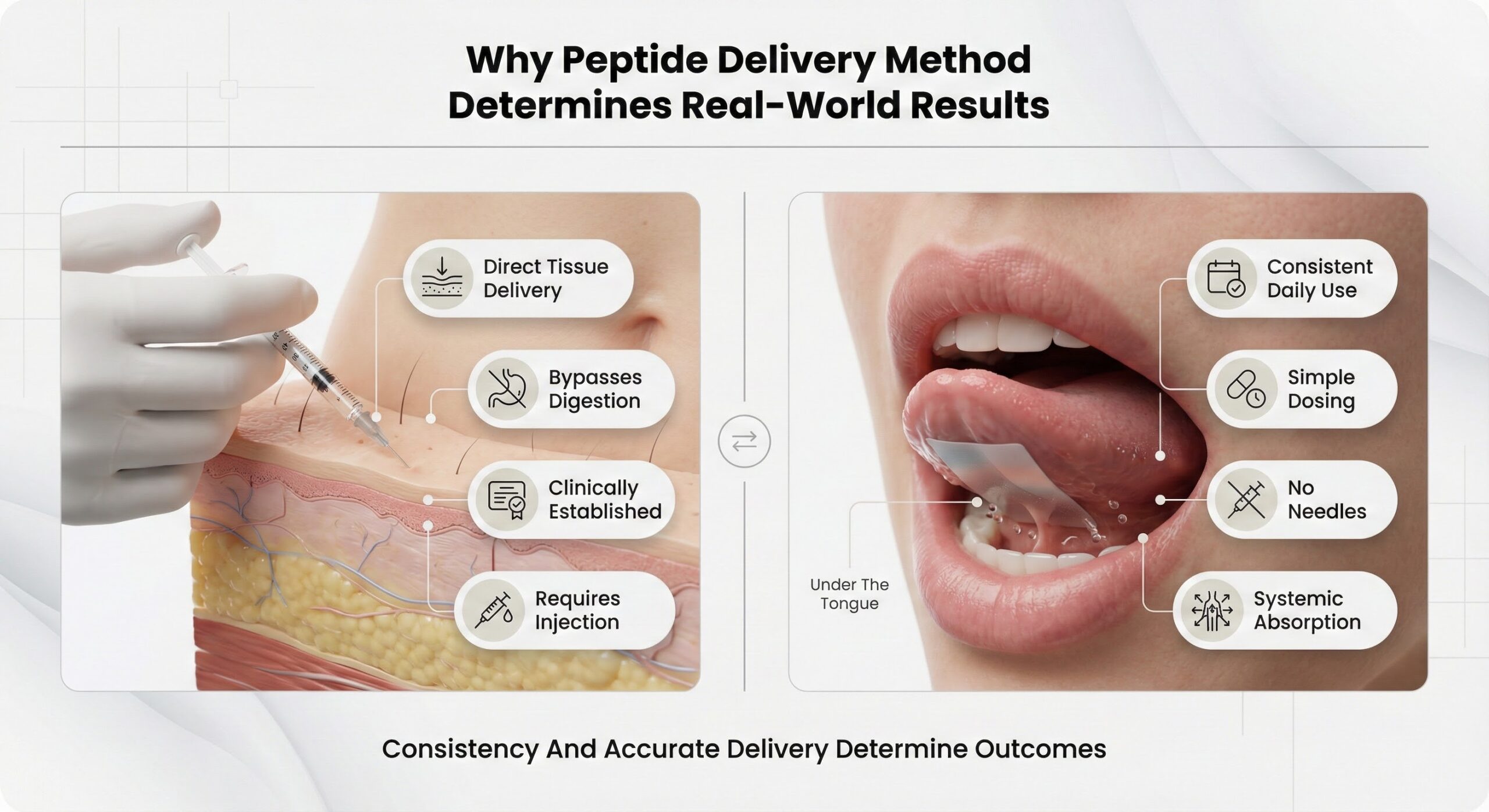
Why Peptide Delivery Method Determines Real-World Results
March 10, 2026