
PT-141 Explained: The Neurological Path to Sexual Wellness
PT-141 addresses sexual dysfunction through direct action on the brain rather than peripheral blood flow. This peptide activates melanocortin receptors MC3R and MC4R in the hypothalamus, modulating neural circuits that generate sexual desire, motivation, and arousal. The result is enhanced libido driven by central nervous system signaling—addressing the cognitive and emotional dimensions of sexual response that vascular treatments cannot reach.[1]

What PT-141 Does for Sexual Function
PT-141 binds to melanocortin receptors concentrated in hypothalamic regions responsible for sexual behavior, triggering neurotransmitter changes that amplify reward pathways, heighten arousal states, and support bonding responses. Studies in healthy men and those with erectile dysfunction showed rapid, dose-dependent increases in erectile activity, with first response occurring within approximately 30 minutes and effects sustained for hours.
In women with hypoactive sexual desire disorder, clinical trials involving over 1,200 participants demonstrated significant increases in satisfying sexual events and meaningful reductions in sexual distress. For men experiencing erectile dysfunction, PT-141 offers benefits by addressing both desire pathways and triggering nitric oxide release in penile tissue. Studies combining PT-141 with sildenafil revealed synergistic effects, with erectile duration increasing more than fivefold compared to sildenafil alone, demonstrating how neurogenic and vascular mechanisms work together.

Why Delivery Method Determines Outcomes
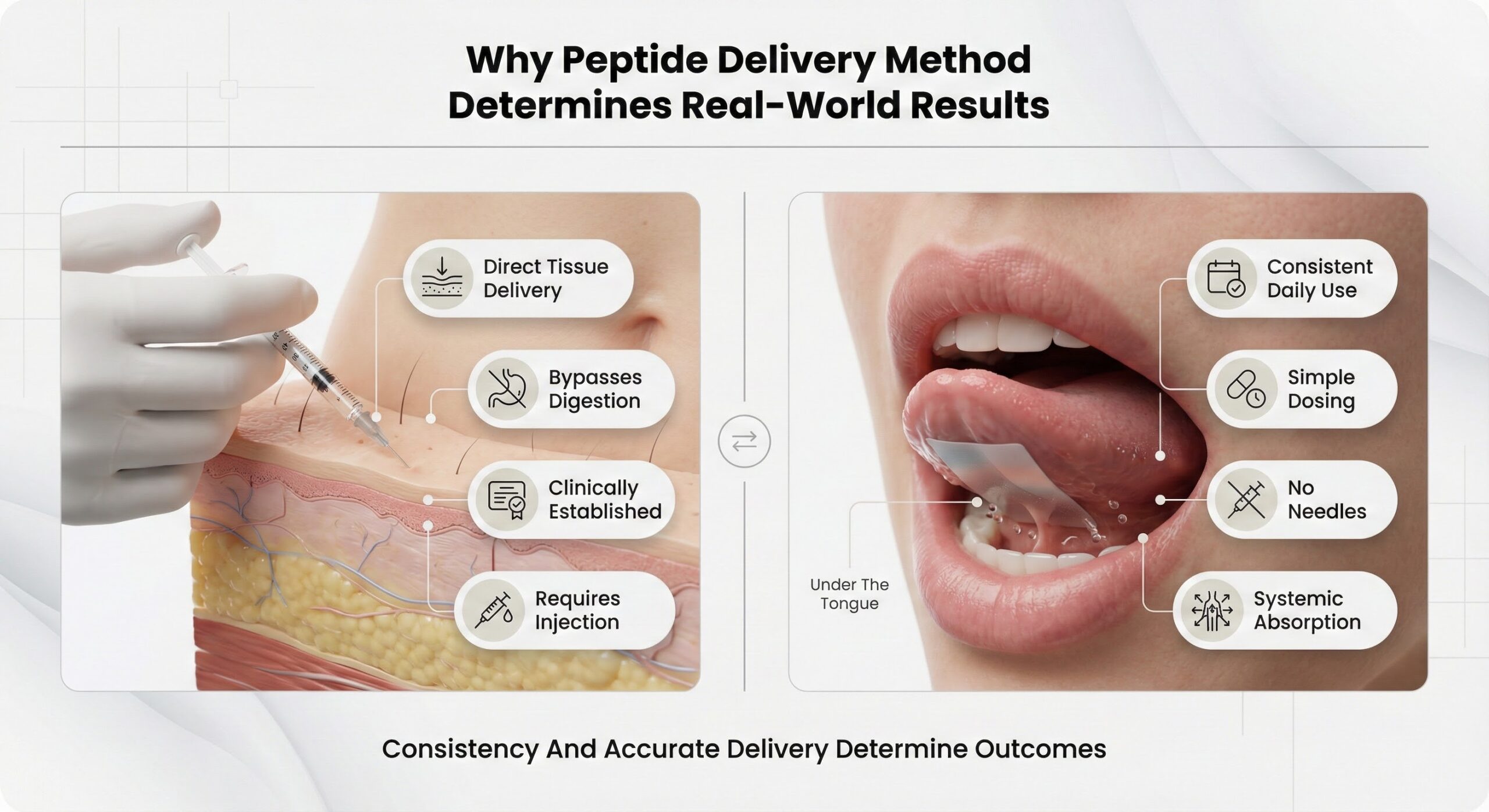
The therapeutic potential of PT-141 depends on whether the peptide reaches melanocortin receptors in functionally active form and whether patients maintain consistent use over time. Subcutaneous injection achieves complete bioavailability with predictable plasma concentrations reached within 30 to 60 minutes. However, injection-based therapy introduces multiple points where effectiveness can break down, independent of the peptide itself.[2]
Reconstitution presents the first challenge, as PT-141 arrives as freeze-dried powder requiring careful mixing with bacteriostatic water. The process demands sterile technique, precise water volume measurement, and gentle mixing to avoid peptide degradation from foaming. Each step creates opportunity for contamination, incorrect concentration, or molecular damage that compromises the dose before administration. Improper reconstitution can lead to denaturation or aggregation of peptides, rendering them ineffective.
Beyond technical preparation, the daily friction of injection therapy erodes adherence. PT-141 works best when used consistently as-needed before sexual activity, but maintaining this pattern requires patients to predict intimate moments, complete multi-step preparation, and overcome injection anxiety. Research shows that logistical issues and injection site complications influence adherence patterns, preventing patients from experiencing the full neurological benefits the peptide can provide.[3]

The Dissolving Strip Advantage
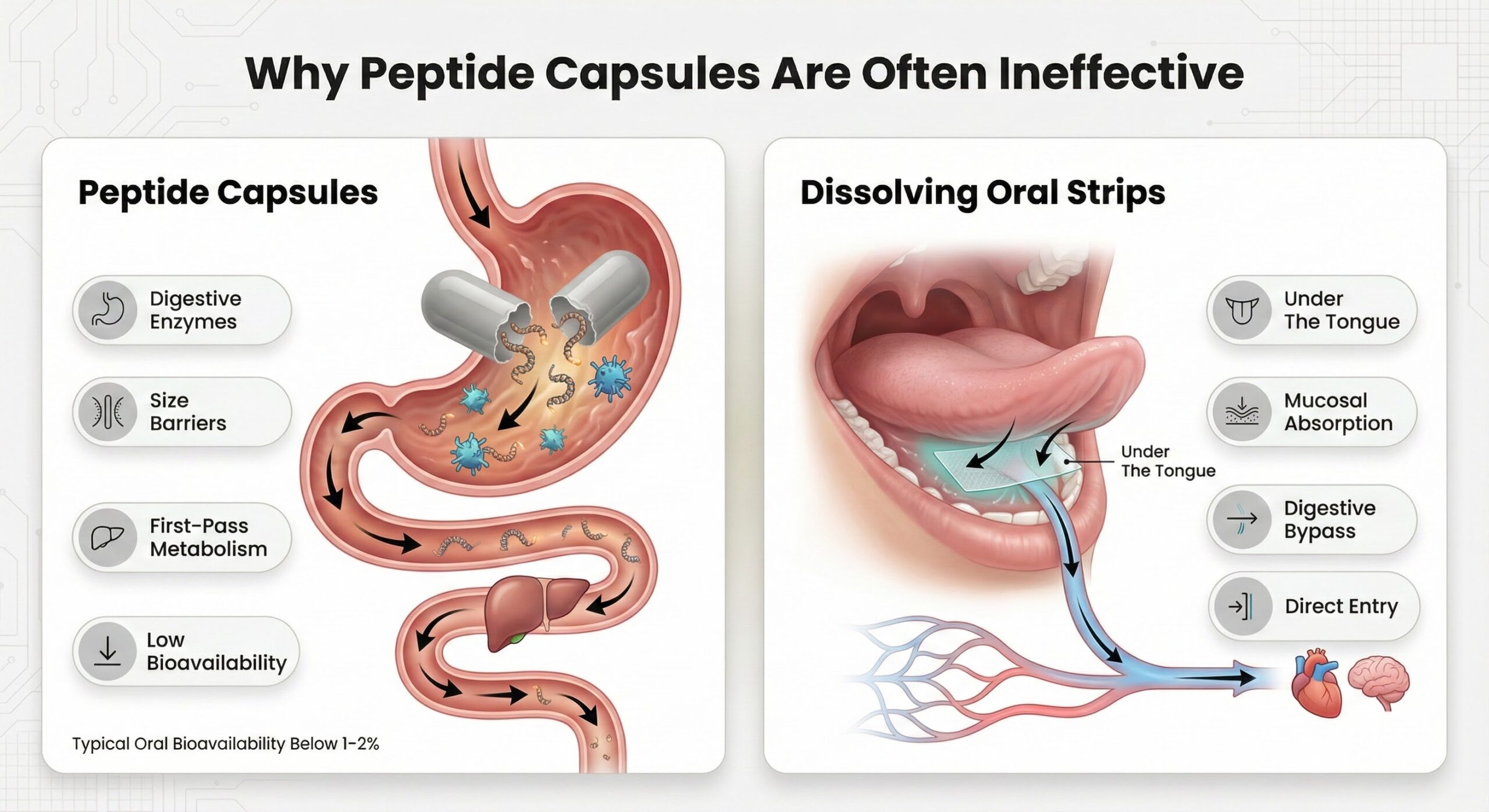
Sublingual strips eliminate the preparation complexity, technical skill requirements, and psychological barriers that undermine injection-based therapy. The peptide is embedded in a pre-measured film that dissolves instantly under the tongue, allowing PT-141 to absorb directly through the rich vascular network of the oral mucosa. This route bypasses first-pass hepatic metabolism while delivering peptides into systemic circulation within minutes.
Sublingual delivery achieves bioavailability rates substantially higher than oral swallowing—studies report sublingual absorption reaching 36.2% compared to just 24.8% for traditional oral routes, with standard oral peptide bioavailability often below 2% due to digestive enzyme breakdown. The thin epithelial membrane beneath the tongue allows peptides to cross directly into capillaries, avoiding the harsh pH fluctuations, enzyme exposure, and intestinal barriers that destroy peptides in the gastrointestinal tract.
Each strip contains an exact, pre-measured dose—no reconstitution calculations, no drawing from vials, no risk of concentration errors. Users simply place the strip under the tongue 30 to 60 minutes before desired activity, with the film designed to dissolve upon contact within seconds. This simplicity translates into dramatically improved adherence: when therapy fits seamlessly into daily routines without physical or psychological friction, patients use it consistently enough to experience sustained benefits.[4]

Making PT-141's Neural Benefits Accessible
PT-141 represents a fundamentally different approach to sexual wellness—one rooted in brain chemistry rather than peripheral blood flow. The peptide's ability to enhance dopaminergic reward circuits, modulate arousal neurotransmitters, and amplify neural responses to erotic stimuli offers meaningful benefits for both women with desire disorders and men seeking libido support. Clinical evidence confirms these neurological effects translate into more satisfying sexual experiences when the peptide is used consistently.
Peptide benefits only materialize when delivery systems support real-world use. Injection therapy imposes preparation complexity, technical demands, and daily friction that progressively erode adherence—the single most important factor in achieving sustained outcomes. Sublingual strips address these execution barriers by providing precise, pre-measured doses in a format that dissolves instantly without needles, mixing, or specialized technique. The result is a delivery system engineered for consistency: easier to use correctly, simpler to maintain over time, and free from the variables that make injection-based protocols difficult to sustain.[3]
References
- Molinoff PB et al. "PT-141: a melanocortin agonist for the treatment of sexual dysfunction." Ann N Y Acad Sci. 2003. [View Study]
- Diamond LE et al. "Double-blind, placebo-controlled evaluation of the safety, pharmacokinetic properties and pharmacodynamic effects of intranasal PT-141, a melanocortin receptor agonist, in healthy males and patients with mild-to-moderate erectile dysfunction." Int J Impot Res. 2004. [View Study]
- Brod M et al. "Understanding compliance issues for daily self-injectable treatment in ambulatory care settings." Patient Prefer Adherence. 2008. [View Study]
- Bala R et al. "Orally dissolving strips: A new approach to oral drug delivery system." Int J Pharm Investig. 2013. [View Study]
- Edinoff AN et al. "Bremelanotide for Treatment of Female Hypoactive Sexual Desire." Neurol Int. 2022. [View Study]
- Kingsberg SA et al. "Bremelanotide for the Treatment of Hypoactive Sexual Desire Disorder: Two Randomized Phase 3 Trials." Obstet Gynecol. 2019. [View Study]
Read More

What GMP Certification Means for Supplement Quality
March 10, 2026
Why Peptide Capsules are Often Ineffective
March 10, 2026
Why Peptide Delivery Method Determines Real-World Results
March 10, 2026





