
Non-Injectable, Non-Pill Delivery for Sexual Health: What Works and Why
Non-pill, non-injectable options can meaningfully support libido, arousal, and comfort through the skin, under the tongue, through nasal tissue, or via local vaginal application—and several of these routes are already backed by human clinical data. Transdermal testosterone has repeatedly improved sexual desire and satisfying sexual events in postmenopausal women with hypoactive sexual desire disorder, while vaginal DHEA restores genital sensitivity and orgasm intensity by converting locally to estrogens and androgens. The practical advantage of these systems is less about stronger effects and more about steady, repeatable exposure with less daily friction than pills or injections, especially when delivery is simplified into fast, under-the-tongue formats.[1]

Why Delivery Route Matters
Non-oral routes like transdermal, sublingual, intranasal, and vaginal delivery bypass first-pass liver metabolism, which increases how much of a compound actually reaches circulation or local tissues. Sublingual absorption, for example, uses the dense capillary network under the tongue to achieve rapid absorption and high systemic availability, often reaching peak levels in about 30 minutes or less for many small molecules. These routes can also change timing dramatically: intranasal sildenafil reaches peak blood levels within approximately 45 minutes (with a Tmax of 0.75 hours), compared to 75–120 minutes for oral tablets, making them better suited for on-demand support. For sexual health, that translates into more predictable arousal, desire, and comfort when the user actually needs it—provided the system is simple enough to use consistently.[2]

Transdermal and Vaginal: The Most Proven
Transdermal testosterone patches at 300 micrograms per day have repeatedly improved sexual desire and increased sexually satisfying events in postmenopausal women with hypoactive sexual desire disorder, with clinical trials showing statistically significant increases in satisfying sexual events compared to placebo (p < 0.0001 to p < 0.05). Testosterone sprays and gels using similar transdermal principles have also shown benefits in women with low sexual interest and low free testosterone, supporting the concept that steady, skin-based delivery can meaningfully affect libido.
Vaginal DHEA (prasterone) has been shown to restore genital sensitivity and improve orgasm intensity by locally converting to estrogens and androgens in vaginal tissues, particularly where estrogen deficiency has caused thinning and discomfort. In a 12-week placebo-controlled trial, intravaginal DHEA improved arousal/sensation by 68% (p = 0.006), orgasm by 75% (p = 0.047), and dryness during intercourse by 57% (p = 0.0001) compared to placebo. These improvements occur exclusively at the vaginal level, without biologically significant changes in serum steroid levels, making it a truly local therapy.[3]

Fast-Acting Intranasal and Brain-Targeted Peptides
Intranasal sildenafil reaches peak blood levels much faster than oral tablets, with human pharmacokinetic data showing a Tmax of 0.75 hours intranasally versus 1.25 hours orally, achieving nearly twofold higher serum drug levels and a relative bioavailability of 112.89%. Intranasal delivery benefits from the rich blood supply in the nasal mucosa and can also provide partial access to central nervous system pathways through olfactory and trigeminal nerve routes, making it a logical route for fast-onset sexual function support where timing is critical.[4]
Intravenous kisspeptin in men with hypoactive sexual desire disorder has been shown to enhance activity in brain regions that process sexual stimuli, increase penile tumescence by 56% more than placebo during erotic visual stimuli, and improve behavioral measures of sexual desire and arousal. This demonstrates how peptide-based approaches can work centrally—modulating sexual motivation and arousal at the brain level—which is highly relevant for future non-injectable formulations that target desire rather than just mechanics.

Where Sublingual Strips Fit In
Sublingual delivery uses the dense capillary network under the tongue to achieve rapid absorption and high systemic availability while bypassing first-pass liver metabolism. Quick-dissolving oral strips take this sublingual principle and remove most user friction: there is no need for water, no swallowing, no measuring, and no remembering complicated instructions—just place, dissolve, and go. Compared with traditional oral capsules, this means less variability from digestion, fewer food interactions, and a more predictable curve of onset and offset—critical when trying to align desire or arousal with real-world intimacy windows.[5]
That simplicity matters because sexual-health outcomes depend on repetition: a delivery system that is easy to use day after day is more likely to produce stable hormonal or neuromodulatory patterns, translating into steadier desire and more reliable sexual experiences over time. Relative to injectables, strips eliminate reconstitution, needle handling, dose-measurement errors, and injection-site reactions, which are all common reasons people skip or delay doses—even when they strongly value the potential benefits. Relative to skin gels or patches, strips avoid issues like variable skin permeability, contact transfer to partners, and the need to manage adhesive sites, making them particularly attractive for users who want central or systemic effects without adding another step to their grooming routine.

Practical Takeaways for Libido Support
For women experiencing hypoactive sexual desire, medically supervised transdermal testosterone or local vaginal DHEA can provide meaningful improvements, especially when combined with lifestyle foundations like resistance training, cardiovascular fitness, stress reduction, and nutrient-dense eating. For timing-sensitive needs, intranasal and sublingual systems offer rapid-onset options that align better with real-life intimacy than slow oral tablets. For ongoing, daily support with peptides or botanicals, dissolving sublingual strips provide a low-friction way to achieve consistent dosing—minimizing user error and complexity so the therapeutic pattern is stable enough for libido benefits to actually show up in day-to-day life.[6]
References
- Ganesan K et al. "Transdermal Testosterone in Female Hypoactive Sexual Desire Disorder: A Rapid Qualitative Systematic Review Using Grading of Recommendations Assessment, Development and Evaluation." Cureus. 2018. [View Study]
- Basson R "Testosterone therapy for reduced libido in women." Ther Adv Endocrinol Metab. 2010. [View Study]
- Labrie F et al. "Effect of intravaginal dehydroepiandrosterone (Prasterone) on libido and sexual dysfunction in postmenopausal women." Menopause. 2009. [View Study]
- Elshafeey AH et al. "Intranasal microemulsion of sildenafil citrate: in vitro evaluation and in vivo pharmacokinetic study in rabbits." AAPS PharmSciTech. 2009. [View Study]
- Bahraminejad S et al. "Sublingual and Buccal Delivery: A Historical and Scientific Prescriptive." Pharmaceutics. 2025. [View Study]
- Labrie F et al. "Effect of Intravaginal Prasterone on Sexual Dysfunction in Postmenopausal Women with Vulvovaginal Atrophy." J Sex Med. 2015. [View Study]
- Crowe TP et al. "Mechanism of intranasal drug delivery directly to the brain." Life Sci. 2018. [View Study]
- Krapf JM et al. "The role of testosterone in the management of hypoactive sexual desire disorder in postmenopausal women." Maturitas. 2009. [View Study]
- Cieri-Hutcherson NE et al. "Systematic Review of l-Arginine for the Treatment of Hypoactive Sexual Desire Disorder and Related Conditions in Women." Pharmacy (Basel). 2021. [View Study]
Read More

What GMP Certification Means for Supplement Quality
March 10, 2026
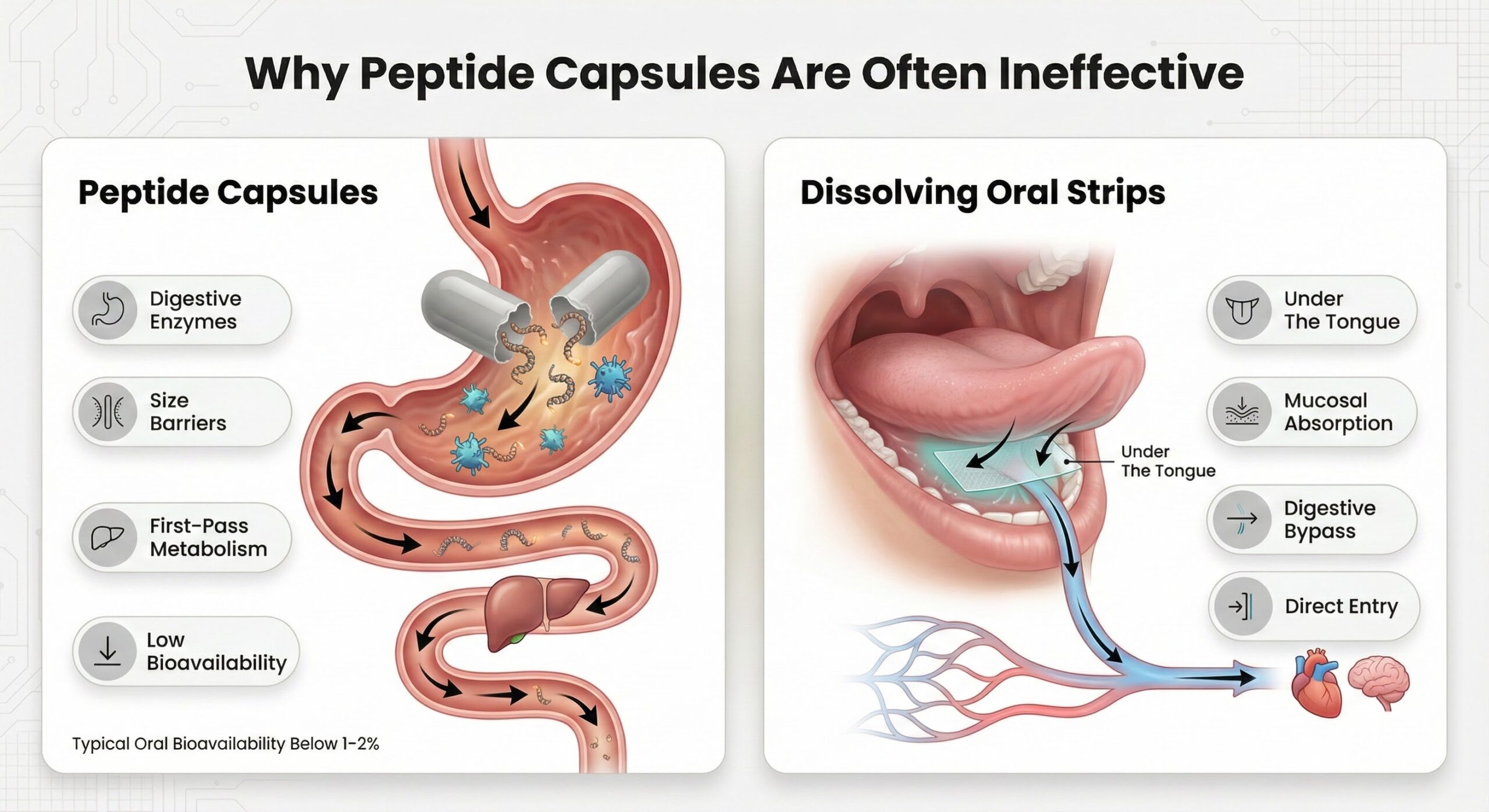
Why Peptide Capsules are Often Ineffective
March 10, 2026
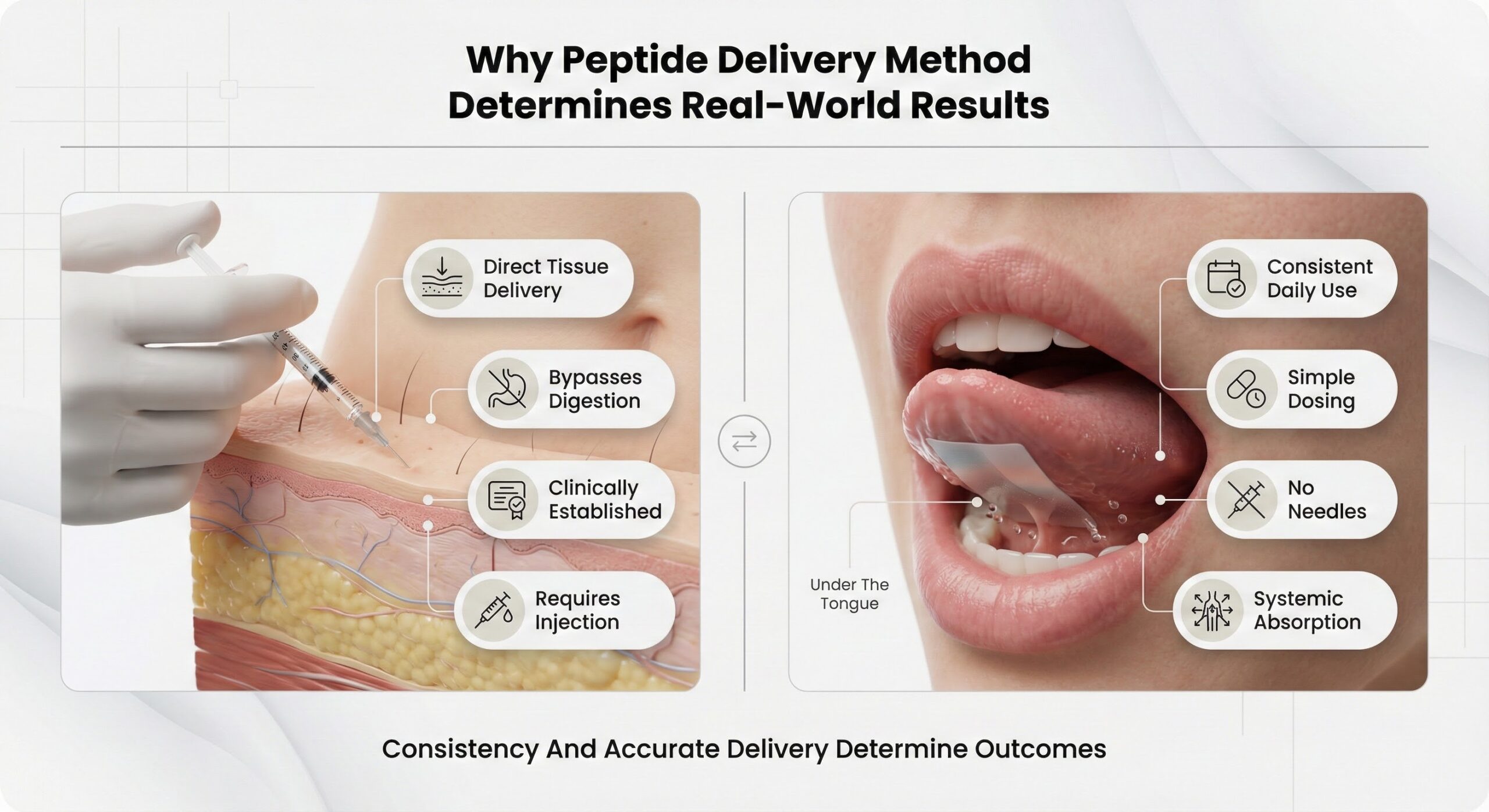
Why Peptide Delivery Method Determines Real-World Results
March 10, 2026





