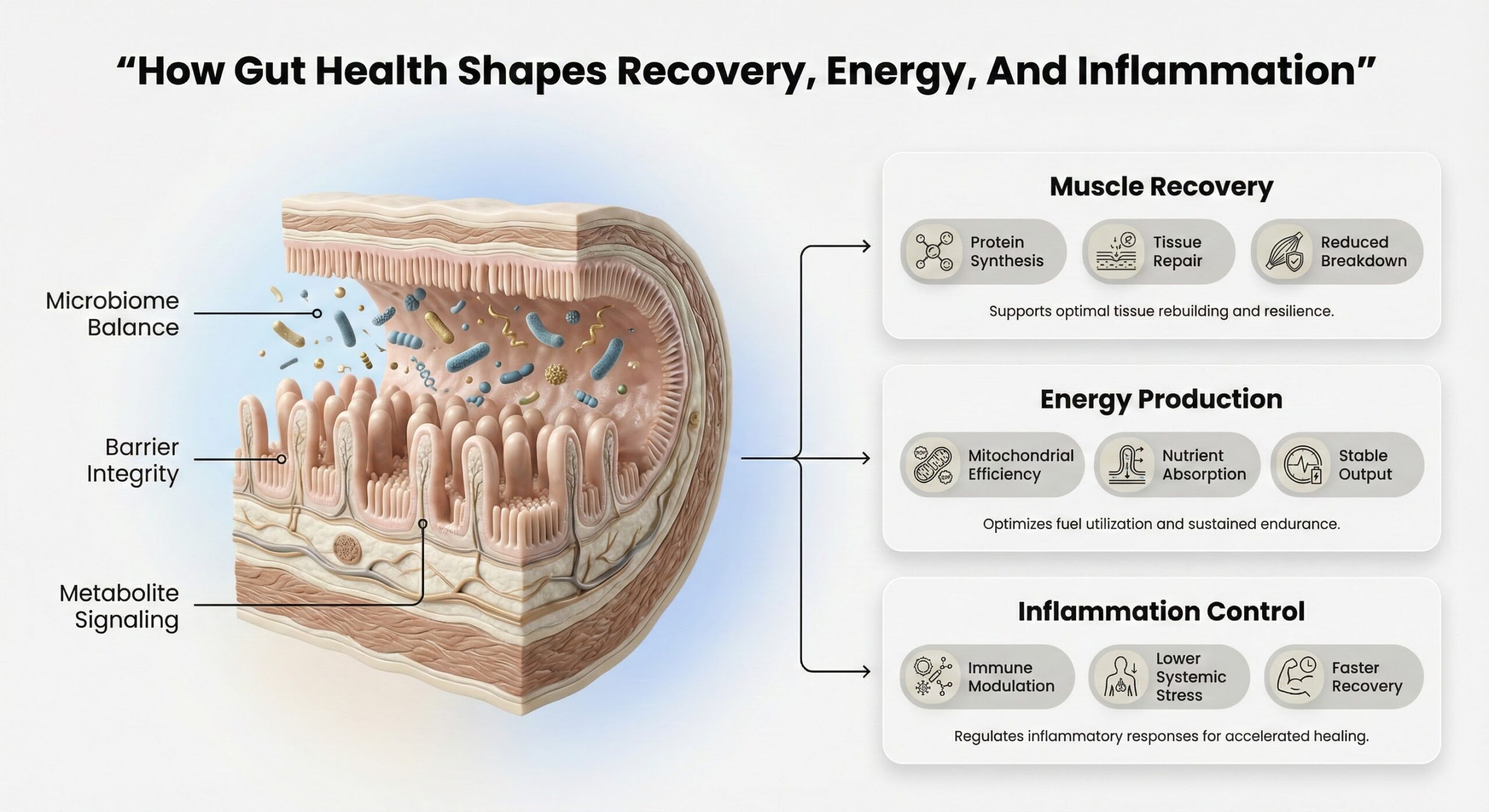
How Gut Health Impacts Recovery, Energy, and Inflammation
Your gut doesn't just digest food—it regulates muscle repair, energy production, and systemic inflammation. The bacteria, metabolites, and barrier integrity of your gastrointestinal system directly influence whether your muscles rebuild after stress, whether your mitochondria produce energy efficiently, and whether chronic inflammation takes hold. These pathways explain why two people with identical training and diet can experience vastly different recovery times, energy levels, and inflammatory responses.
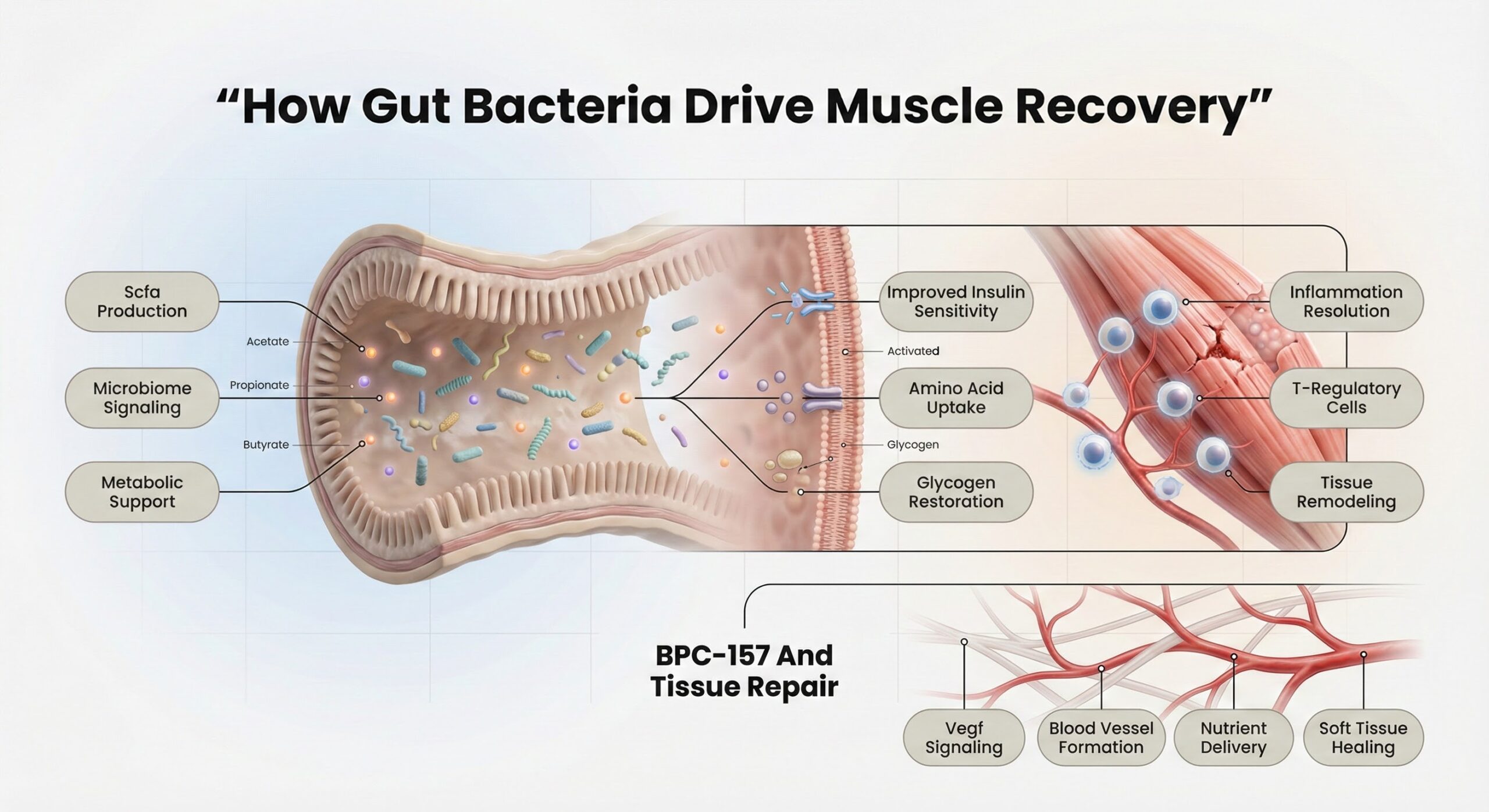
How Gut Bacteria Drive Muscle Recovery
Muscle repair depends on whether your body can signal tissue reconstruction and deliver nutrients to damaged fibers. Gut bacteria produce short-chain fatty acids (SCFAs) like acetate, propionate, and butyrate, which bind to receptors on muscle cells and promote insulin sensitivity, allowing amino acids and glucose to enter tissues more efficiently. This process restores glycogen stores and initiates anabolic signaling after exercise or injury.[1]
Beyond metabolic support, the gut microbiome regulates immune modulation during recovery. After tissue damage, the body must transition from inflammation to remodeling. Gut-derived SCFAs suppress excessive inflammatory responses by promoting T-regulatory cells, which migrate to injured sites and prevent prolonged tissue breakdown. This shift separates effective recovery from chronic soreness or delayed healing.
BPC-157, a peptide derived from gastric proteins, accelerates soft tissue repair by upregulating vascular endothelial growth factor (VEGF), which promotes blood vessel formation and increases nutrient delivery to tendons, ligaments, and muscle tissue. Unlike most peptides, BPC-157 remains stable in gastric juice for over 24 hours, supporting consistent daily use through oral delivery.
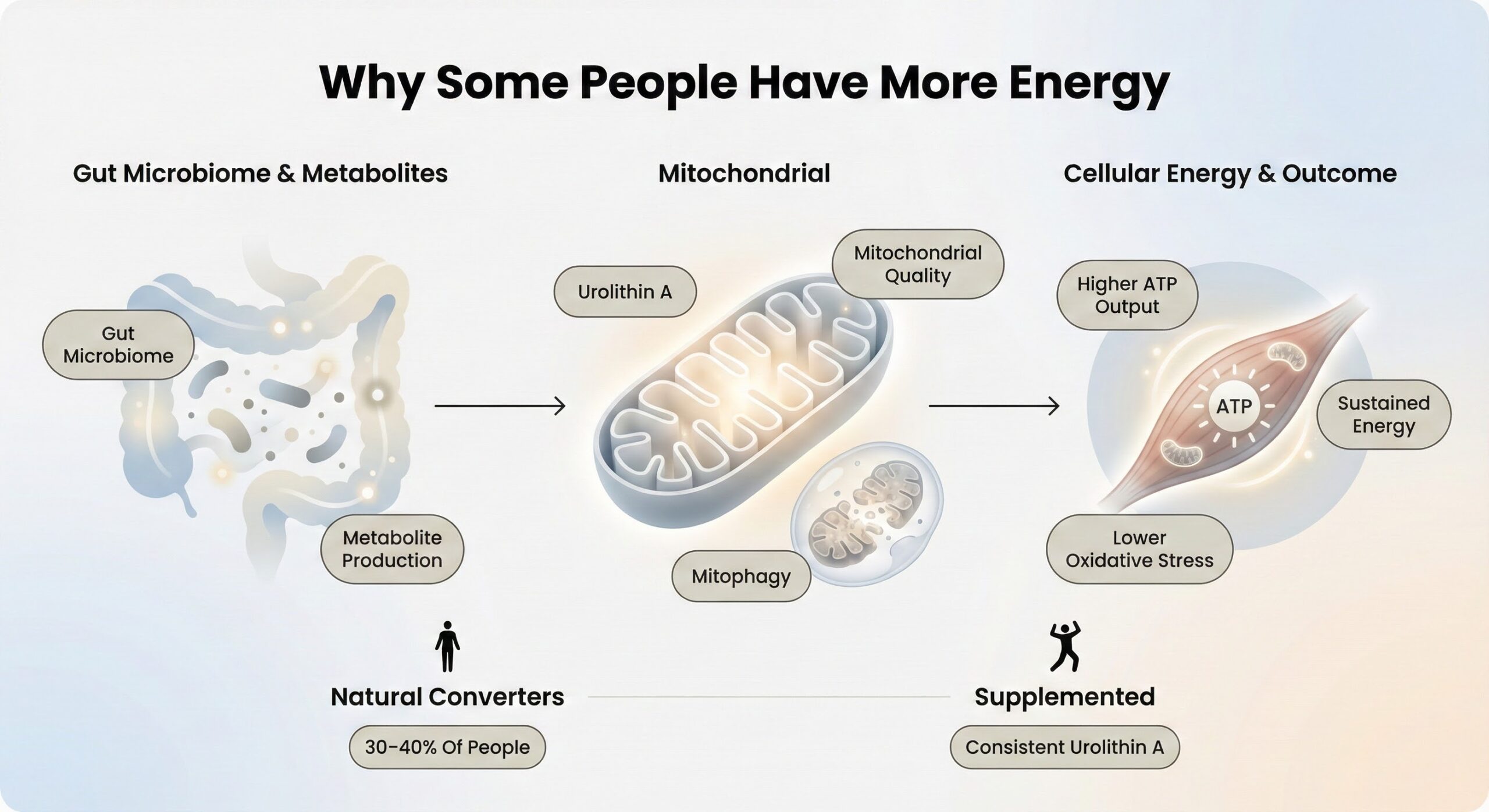
Why Some People Have More Energy
Fatigue isn't always about sleep or calories—it's often about mitochondrial efficiency. Your gut microbiome produces metabolites that directly regulate how well your cells generate ATP, the energy currency of the body. Urolithin A, a compound produced when gut bacteria metabolize ellagitannins from pomegranates and berries, activates mitophagy—the selective removal of defective mitochondria. This process ensures that only high-functioning mitochondria remain active, improving cellular energy output and reducing oxidative stress.
In older adults taking 1,000 mg of urolithin A daily for four months, muscle endurance improved significantly, measured by the number of contractions until fatigue in both hand and leg muscles. Plasma biomarkers associated with mitochondrial health, including acylcarnitines and ceramides, also improved, indicating enhanced energy metabolism at the cellular level.
Only about 30-40% of people naturally convert dietary sources into urolithin A due to gut bacteria differences. Direct supplementation delivers consistent results regardless of baseline microbiome status.
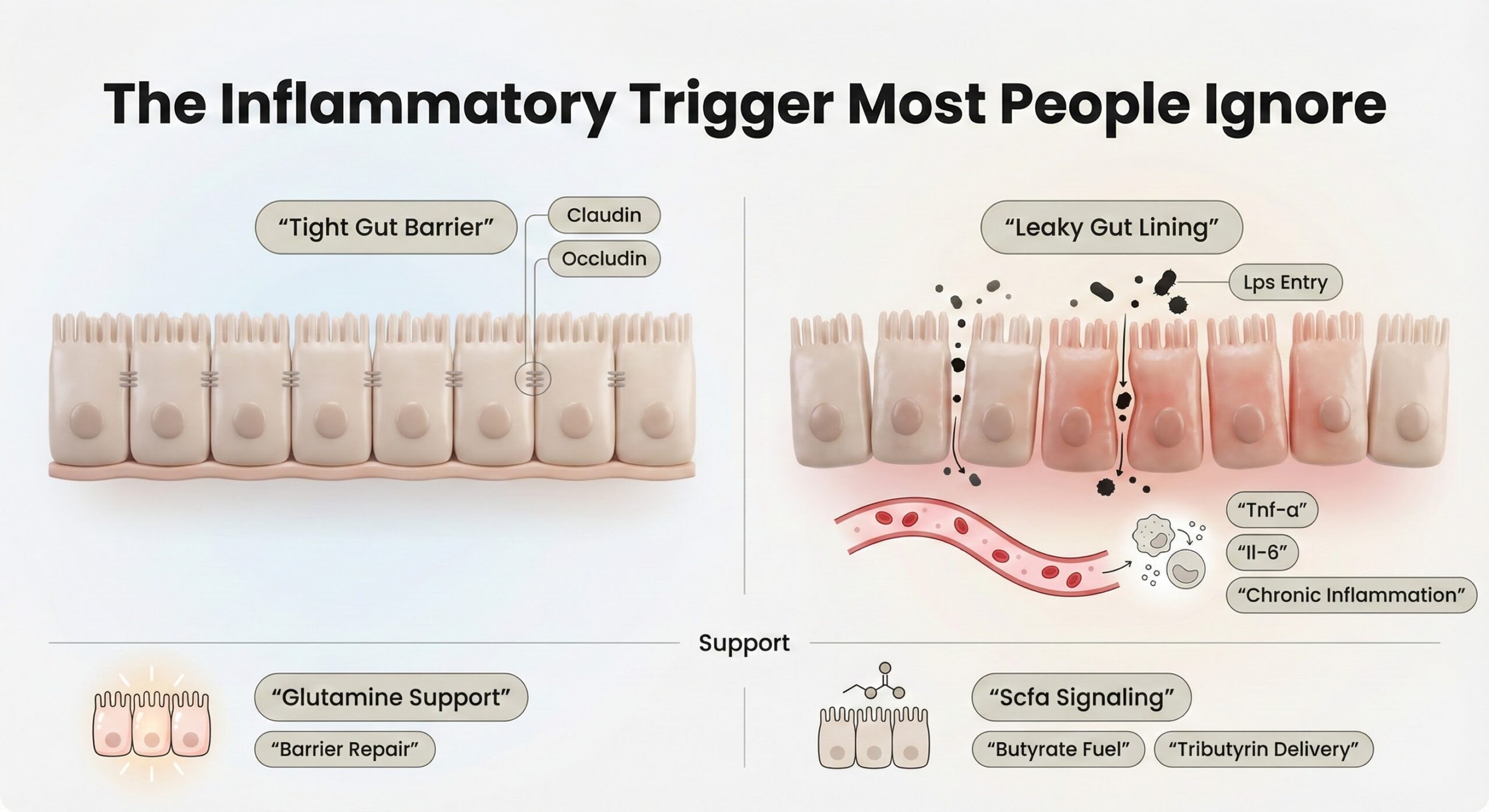
The Inflammatory Trigger Most People Ignore
Chronic inflammation often begins with a compromised gut lining. The intestinal epithelium is held together by tight junction proteins like claudin and occludin, which prevent bacterial toxins and undigested food particles from entering the bloodstream. When these junctions weaken, lipopolysaccharides (LPS) from gram-negative bacteria enter circulation, triggering pro-inflammatory cytokines such as TNF-α and IL-6. This drives systemic inflammation linked to metabolic syndrome, autoimmune flare-ups, and prolonged muscle soreness.
Glutamine, the primary fuel source for intestinal cells, maintains barrier integrity. Supplementation restores tight junction function after ischemia-reperfusion injury, reduces permeability caused by nonsteroidal anti-inflammatory drugs (NSAIDs), and prevents bacterial translocation. Therapeutic doses typically range from 5 to 20 grams daily.
SCFAs also contribute to barrier function by inhibiting histone deacetylases (HDACs), effectively silencing genes responsible for chronic inflammatory responses. Butyrate provides approximately 70% of the energy for colonocytes and helps regulate the immune environment of the gut lining. Esterified forms like tributyrin improve stability and ensure delivery to target tissues.[2]
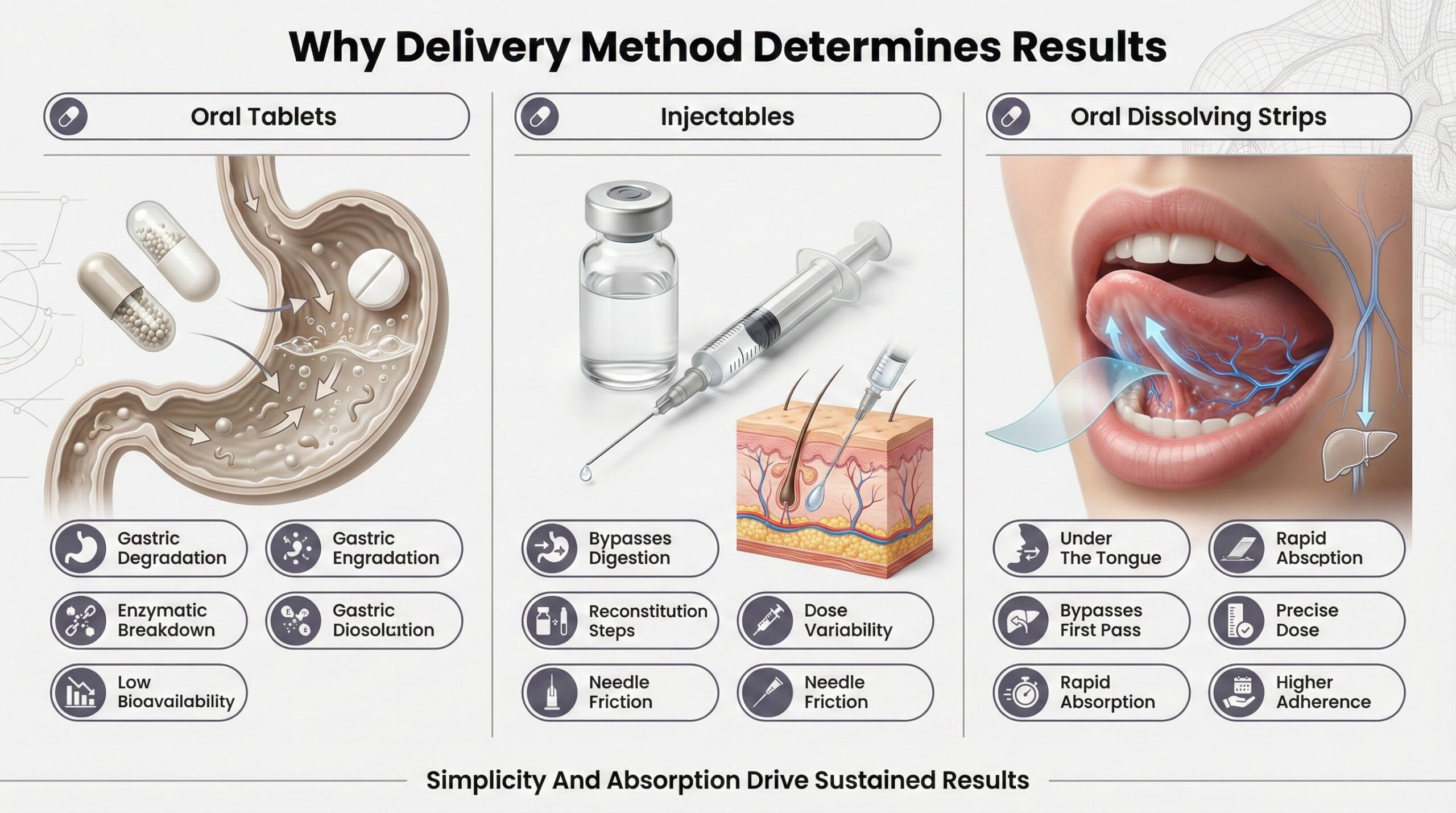
Why Delivery Method Determines Results
The mechanisms above depend on whether active compounds reach systemic circulation or target tissues in functional form. Peptides and metabolites are inherently unstable in the digestive environment, where gastric acid, proteolytic enzymes, and hepatic first-pass metabolism rapidly degrade them.
Oral tablets and capsules face bioavailability challenges due to enzymatic breakdown in the stomach and intestines. Injectable peptides bypass this but introduce complexity—reconstitution steps, measurement variability, injection-site risks, and psychological resistance to needles create friction points that reduce long-term adherence.
Oral dissolving strips deliver compounds through the sublingual mucosa, bypassing both gastric degradation and hepatic first-pass metabolism. The sublingual route allows direct absorption into systemic circulation, with bioavailability rates significantly higher than traditional oral routes. The large surface area of the oral mucosa facilitates rapid dissolution and absorption within seconds.
Beyond pharmacokinetics, dissolving strips eliminate execution errors that plague both injectables and traditional oral supplements. There's no need for mixing, measuring, or injection technique—each strip contains a precise dose that dissolves on contact with saliva. This simplicity translates to better adherence, which is the single most important factor in whether any intervention produces sustained results.
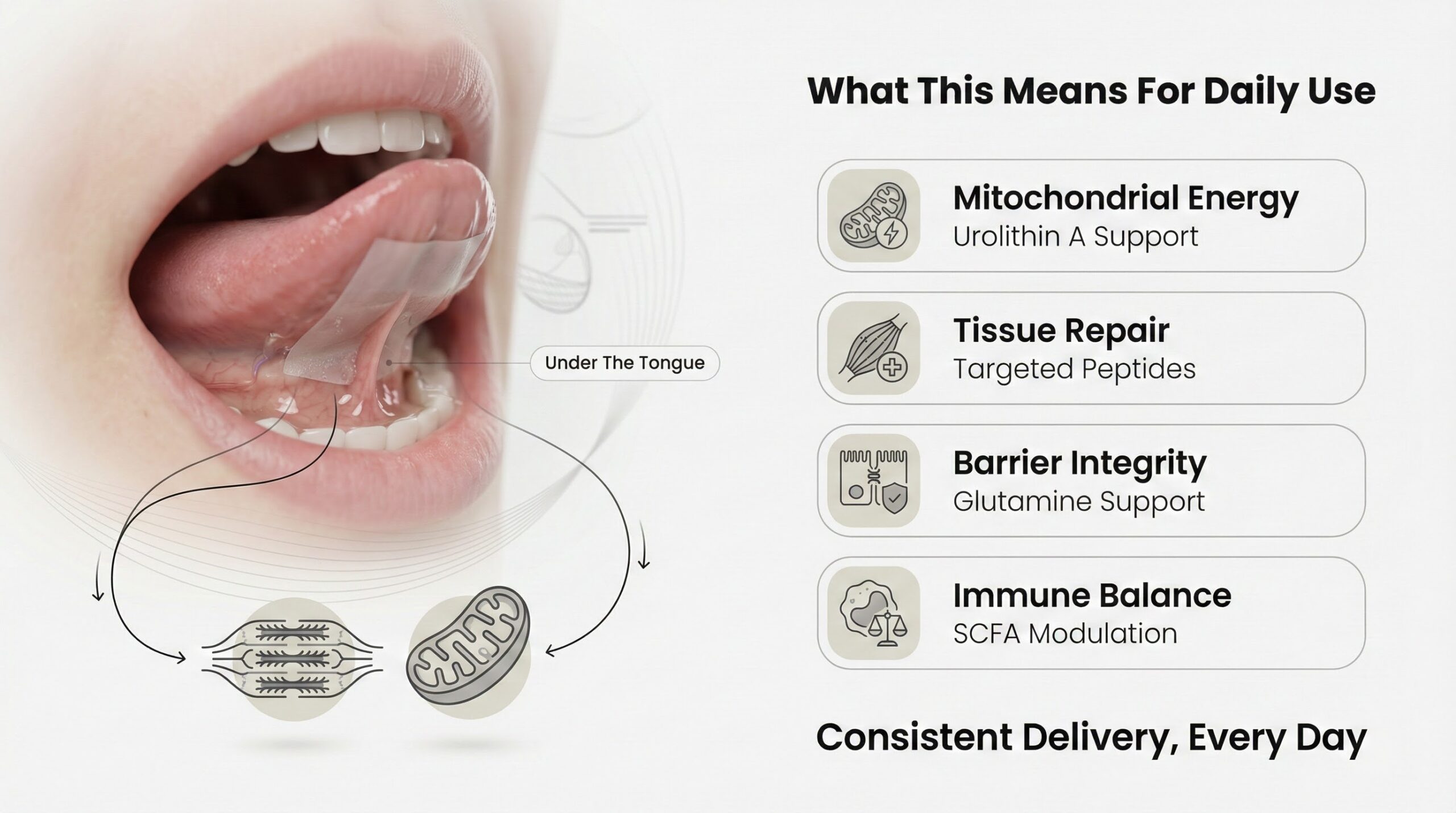
What This Means for Daily Use
If you're recovering slowly, feeling fatigued despite adequate rest, or dealing with persistent inflammation, the issue may be that your gut microbiome isn't producing the metabolites your muscles and mitochondria need, or that your intestinal barrier is allowing inflammatory triggers into circulation. The interventions that support these pathways—urolithin A for mitochondrial function, targeted peptides for tissue repair, glutamine for barrier integrity, and SCFAs for immune modulation—only work if they reach their targets in active form.
Dissolving oral strips make these mechanisms accessible without the complexity of injectables or the inconsistency of traditional oral forms. For anyone prioritizing recovery, sustained energy, and reduced inflammation, the question isn't whether gut-based pathways matter—it's whether you're using a delivery system that lets you experience those benefits reliably, every day.
References
- Liu X et al. "Role and Mechanism of Short-Chain Fatty Acids in Skeletal Muscle Homeostasis and Exercise Performance." Nutrients. 2025. [View Study]
- Steliou K et al. "Butyrate histone deacetylase inhibitors." Biores Open Access. 2012. [View Study]
- Zhang P "Influence of Foods and Nutrition on the Gut Microbiome and Implications for Intestinal Health." Int J Mol Sci. 2022. [View Study]
- [View Study]
- Liu S et al. "Effect of Urolithin A Supplementation on Muscle Endurance and Mitochondrial Health in Older Adults: A Randomized Clinical Trial." JAMA Netw Open. 2022. [View Study]
- Rao R et al. "Role of Glutamine in Protection of Intestinal Epithelial Tight Junctions." J Epithel Biol Pharmacol. 2012. [View Study]
Read More

Preventing Overuse Injuries: The Role of Recovery
March 11, 2026

What GMP Certification Means for Supplement Quality
March 10, 2026
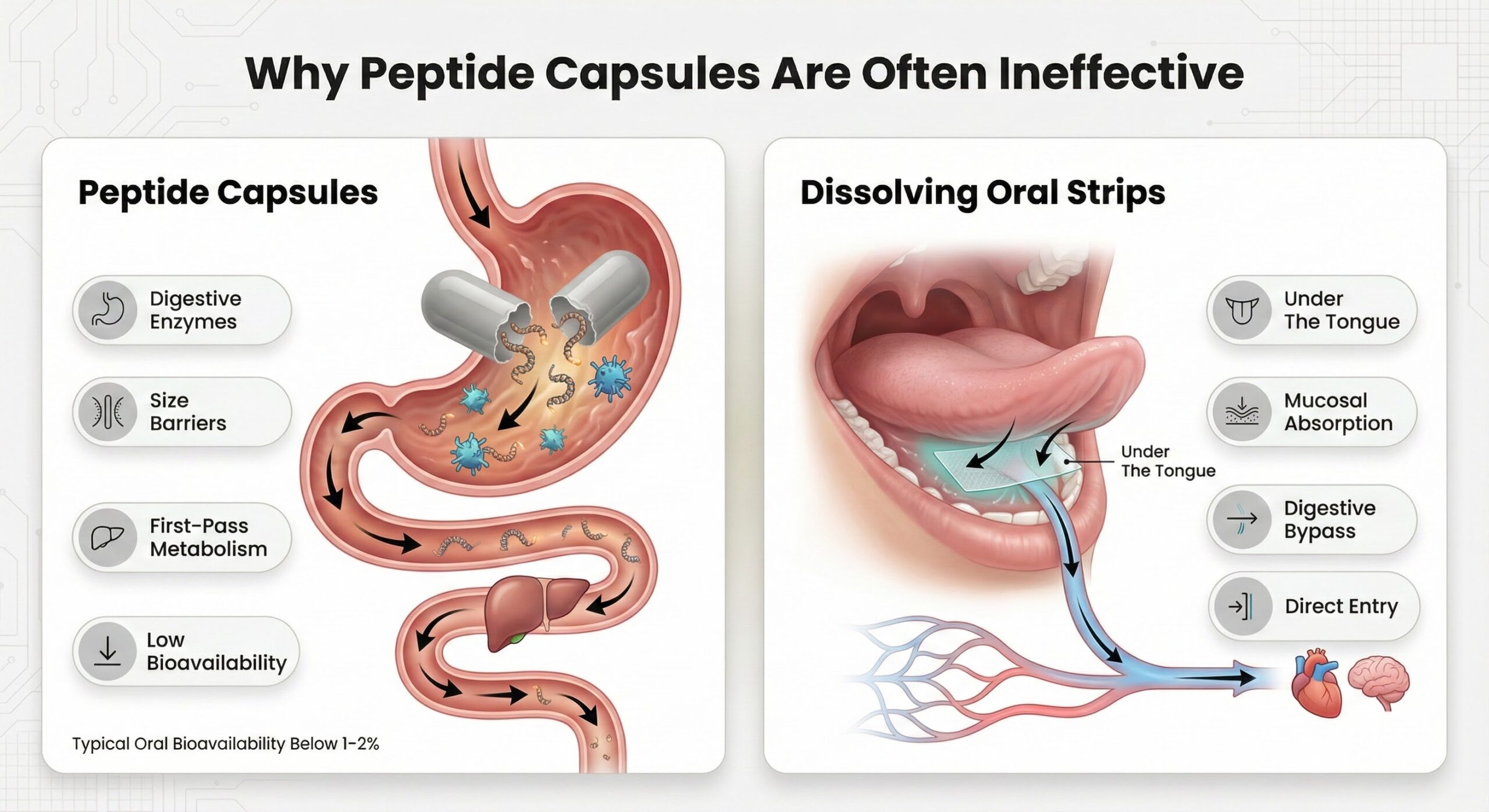
Why Peptide Capsules are Often Ineffective
March 10, 2026





