
Why Manufacturing Consistency Determines Peptide Results
Peptides deliver measurable benefits for tissue repair, recovery acceleration, and inflammatory regulation when used consistently over time. The reliability of those outcomes depends on precise dosing, stable ingredient composition, and elimination of variables that compromise daily adherence. Manufacturing controls that ensure batch-to-batch consistency and reduce user error directly impact whether peptide benefits translate from potential to experienced reality.
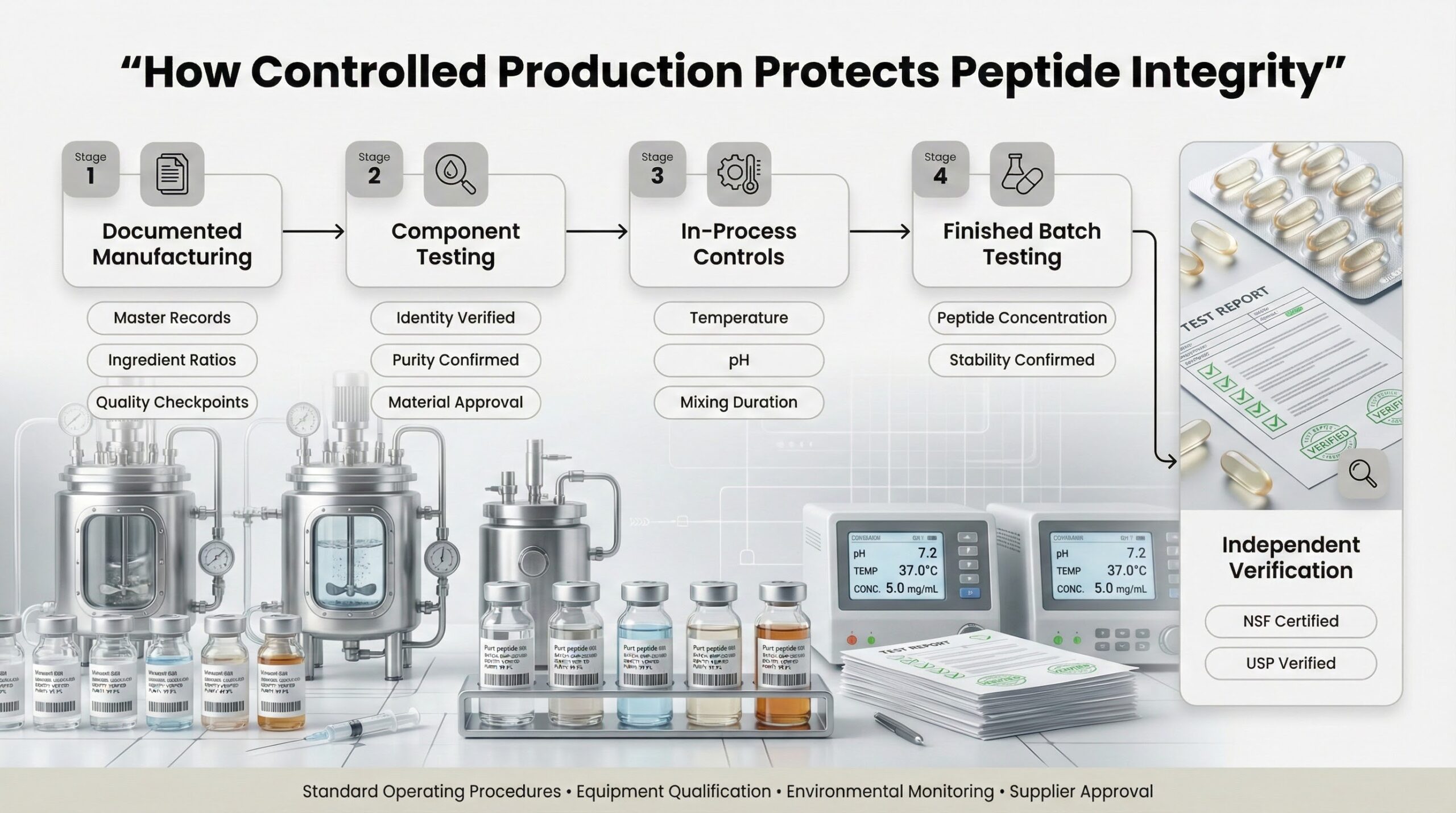
How Controlled Production Protects Peptide Integrity
GMP-certified manufacturing facilities document every production step through master manufacturing records that specify ingredient ratios, processing conditions, and quality checkpoints. Component testing verifies the identity, purity, and composition of incoming peptide materials before they enter production. In-process controls monitor critical parameters—temperature, pH, mixing duration—at stages where deviations would compromise finished product quality. Finished batch testing confirms that each production run meets predetermined specifications for peptide concentration and stability.
Third-party certification from organizations like NSF or USP provides independent verification that these quality systems function as documented. Certified facilities maintain standard operating procedures for equipment qualification, environmental monitoring, and supplier approval—creating traceable accountability for every material that contacts the final product. This systematic approach prevents the cross-contamination, measurement error, and ingredient variability that cause inconsistent results in uncontrolled production environments.

Delivery Method as Part of Manufacturing Design
The way peptides reach systemic circulation affects both bioavailability timing and day-to-day consistency. Oral dissolving strips manufactured under GMP protocols achieve therapeutic plasma concentrations within 10-30 minutes through sublingual absorption, delivering peak levels comparable to injection-based methods. The controlled release profile sustains peptide presence above therapeutic thresholds for several hours, reducing the need for multiple daily doses.
More critically, strips eliminate the multi-step preparation system that introduces variability in traditional delivery: no reconstitution measurements, no injection-site rotation decisions, no refrigeration transport requirements. Each strip contains a pre-measured peptide dose manufactured to specification under documented controls—the user simply places it sublingually and allows absorption to proceed. This reduction in preparation complexity removes failure points where measurement error, contamination, or inconsistent technique compromise intended dosing.

Manufacturing Controls That Support Daily Use
GMP certification verifies that production facilities maintain quality management systems with clear responsibility chains, validated processing methods, and documented deviation investigations. Batch records create traceability from raw material receipt through finished product release, enabling manufacturers to identify and correct process variations before they affect multiple production runs.
For peptide users, these controls translate to predictable composition across every strip in a package and consistency between reorders months apart. Stability testing conducted under GMP protocols determines how environmental factors affect peptide degradation over shelf life, establishing storage parameters that maintain potency until the labeled expiration date. The result is a delivery system where the peptide dose consumed on day one matches the dose consumed on day ninety—essential for protocols requiring sustained daily use to produce cumulative benefits.

Why Simplified Execution Improves Outcomes
Adherence to daily peptide protocols directly influences whether users experience the tissue repair, recovery, and inflammatory benefits documented in clinical contexts. Delivery methods requiring multi-step preparation, measurement decisions, or administration discomfort create friction that reduces compliance over weeks and months. Oral dissolving strips remove that friction entirely: the manufacturing facility controls dosing precision, the sublingual route bypasses first-pass metabolism, and the user simply maintains a daily routine without preparation burden.
GMP-certified strip production ensures that this simplified execution doesn't sacrifice consistency—documented process controls, validated equipment, and batch testing mean every strip delivers the intended peptide dose with the same reliability as more complex delivery systems. The difference lies in where complexity exists: in a controlled manufacturing environment with trained personnel and quality oversight, rather than in a user's daily routine where variability accumulates. This architectural shift—from user-dependent preparation to manufacturer-controlled dosing—makes consistent peptide benefits accessible through simple daily use.
Read More

Preventing Overuse Injuries: The Role of Recovery
March 11, 2026

What GMP Certification Means for Supplement Quality
March 10, 2026
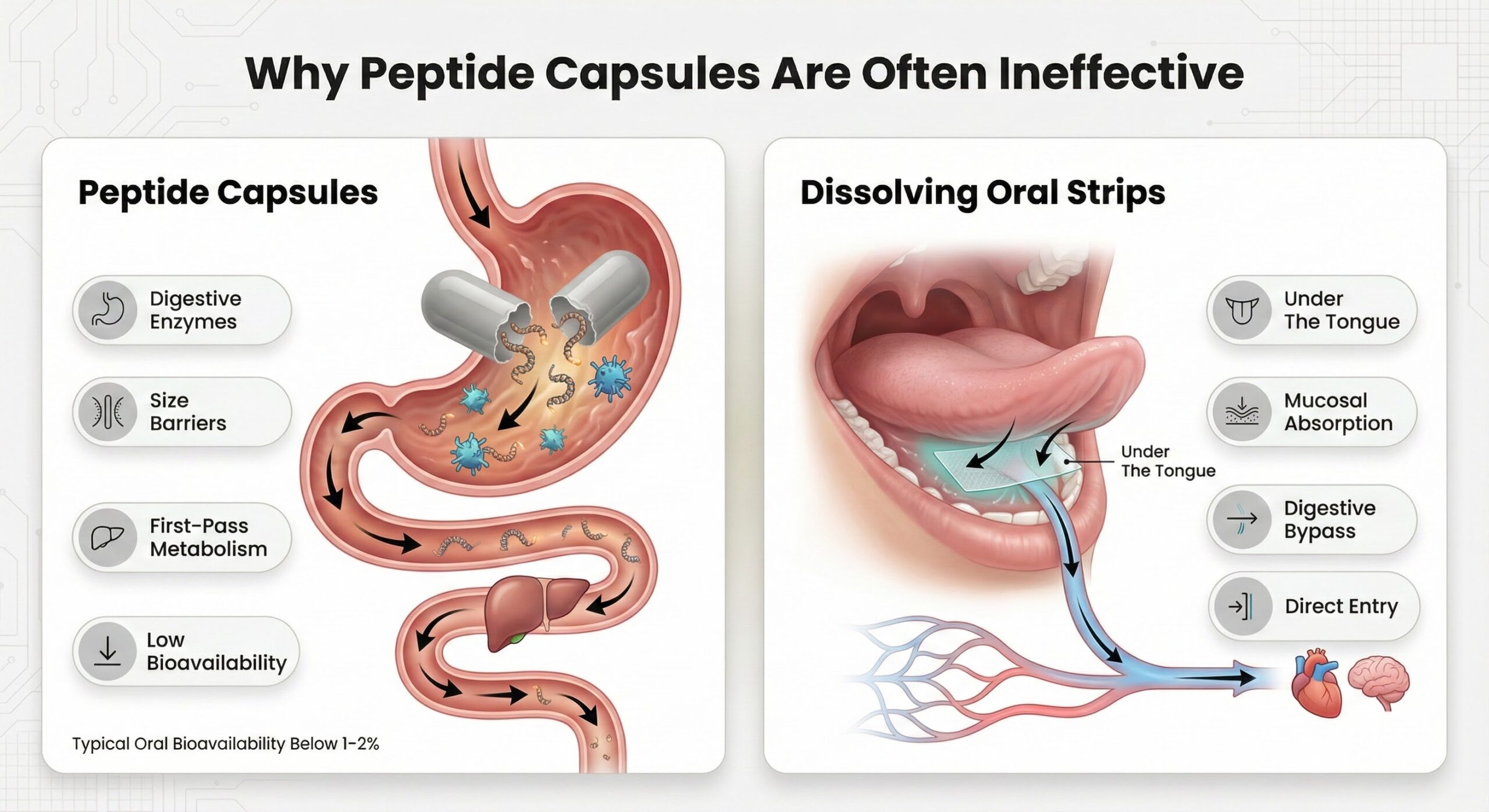
Why Peptide Capsules are Often Ineffective
March 10, 2026





